 But atoms are the smallest particle to take part in a chemical reaction.Īccording to one postulate, the masses and densities of two different elements must differ. Like Isotopes of Hydrogen are deuterium and tritium, their masses are 1,2 and 3, respectively. But isotopes of the same element have different atomic masses. This proved that atoms were further divisible and were not the basic unit of matter.Īs one of the postulates states, the masses and densities of an element should be the same. 
These were protons, neutrons, and electrons. Later after the postulation of Dalton’s atomic theory, subatomic particles were discovered. The theory doesn’t account for subatomic particles.they are always in a fixed ratio concerning their mass. Law of constant composition: In a pure compound, the proportion of elements remains constant, i.e.That is because mass can neither be created nor be destroyed in a chemical reaction. Law of conservation of mass: This law states that in a chemical reaction, the mass of an element before and after the reaction is the same.Atoms can be combined, rearranged, or separated in a chemical reaction.ĭalton’s theory was based on the following concepts that other scientists coined.To form compounds, elements of different atoms can combine in fixed ratios that too in whole-number ratio.They are not further divisible into smaller particles. Atoms can neither be created nor be destroyed.However, different elements exhibit different atoms with different properties and masses and sizes. A particular element consists of all kinds of atoms with the same mass, size, and other properties.All matter is made up of microscopic, indivisible particles known as atoms. 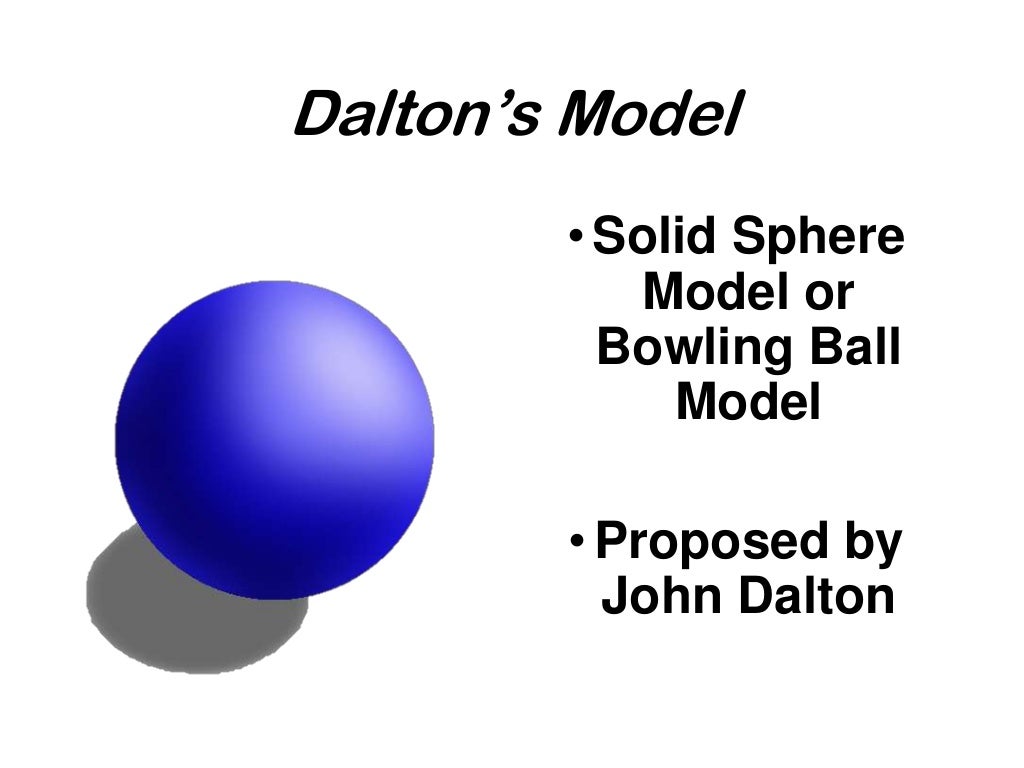
But soon after, the model was discarded due to various demerits of Dalton’s atomic theory. All atoms of a particular element are the same in shape and size, whereas they differ from one element to another. Atoms are indivisible, and the structure of atoms is indestructible, and they are the building units forming all the substances. Dalton was a chemist and an English physicist who put forward his theory on the nature of matter in 1808. One of the initial theories to be given to explain this was called Dalton’s atomic theory. The scientific theory that all matter is made up of atoms is the atomic theory. But his significant contribution to the field of science was the atomic theory based on Dalton’s structure of the atom, also called the Billiard ball model. 
He also defined some significant concepts of partial pressure in physics. He also contributed to Manchester’s literary and philosophical society by publishing a paper on colour blindness called Daltonism for some time. Thus, he was also known as the father of meteorology in some circles. This book created a significant shift in meteorology from folklore to a serious scientific pursuit of the subject. His work, Meteorological Observation, and Essays contained many original ideas of his and his friends and was published in 1793. He was born in England on September 6, 1766. Atomic theory was first proposed by John Dalton, an English physicist and a chemist of the 19th century. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
